Mysteries of the Hearing Brain – Is It ANSD or Cortical Deafness?
Two types of auditory disorders can easily be missed when the audiologic evaluation is limited to assessments of the peripheral auditory system. One disorder, auditory neuropathy spectrum disorder (ANSD), should ordinarily be detected when auditory brainstem response (ABR) testing is used in infant screening. But, ANSD can occur in infants who have no risk factors, and it will not be detected by programs that use otoacoustic emissions (OAE) screening for healthy low-risk infants. Because some individuals with ANSD have completely normal audiograms, this disorder can go undiagnosed well into childhood or adulthood. The chief complaint of individuals with ANSD and a normal audiogram would be difficulty hearing in noise. A key feature of ANSD is a disruption of neural synchrony,1, 2 and previous studies have demonstrated the disruptions in neural synchrony are associated with hearing difficulties in noise.3, 4 Therefore, it is important to include tests of word or sentence recognition in noise in the clinical protocol, especially when the patient expresses this particular complaint. Scores on hearing-in-noise tests that are considerably worse than would be predicted from the audiogram are an indicator of the need to perform follow-up testing that can assess deficits beyond the cochlea. The auditory brainstem response, recorded with both rarefaction and condensation polarities, should show an extended cochlear microphonic in individuals with ANSD (Figure 1).
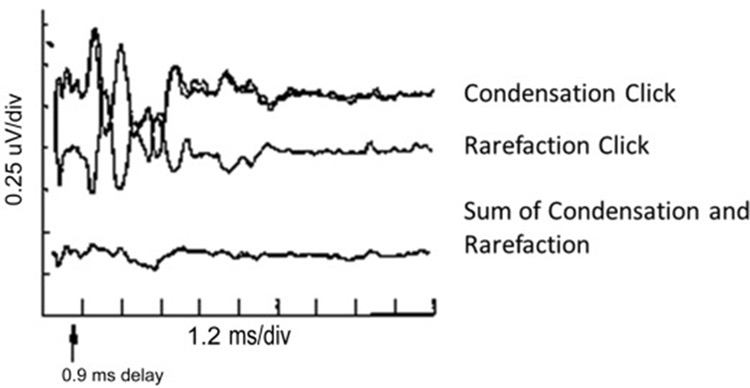
Figure 1. Auditory brainstem response waveforms obtained to clicks presented with condensation and rarefaction polarities demonstrate the reversal of polarity that is characteristic of auditory neuropathy spectrum disorder. Summing the two polarities cancels these components, demonstrating the lack of a true neural response. https://entokey.com/auditory-neuropathydys-synchrony-disorder/
The second type of disorder, cortical deafness, may be confused with nonorganic hearing loss. Cortical deafness is much rarer than ANSD and occurs when there are bilateral lesions in the primary auditory cortex. If you see a patient that has suffered a stroke, cortical deafness is one possible complication. The individual's responses may be inconsistent for pure-tone testing, yet otoacoustic emissions and auditory brainstem responses are normal. The individual with cortical deafness may report being “deaf.” The middle latency response is expected to be absent5.
A recently published case study contrasted two patients, one patient diagnosed with ANSD and the other with cortical deafness.6 The etiology of the patient with ANSD is unknown. Despite her complaints of difficulty hearing, her symptoms were repeatedly dismissed because of normal audiograms. ANSD was not diagnosed until she reached the age of 18 when an ABR revealed absent responses. It was quite a relief to her to have her experiences validated. I often present this case to my students as an example of the potential for misdiagnosis when our audiological evaluation focuses on audiometric thresholds and word recognition in quiet. The patient with cortical deafness experienced bilateral auditory cortex lesions following treatment for acute lymphoblastic leukemia. His symptoms are quite apparent in that he lacks awareness of sound to the extent that he cannot respond consistently to a pure-tone audiogram. At the time of testing, all communication with him was in written form. He has normal distortion-product OAEs and a normal ABR, but he has absent cortical responses.
Figure 2 displays frequency-following responses (FFRs) and cortical responses in these two patients. Once we can see that the patient with cortical deafness has a robust FFR and an absent cortical response. The patient with ANSD has a largely absent/minimal FFR but a relatively robust cortical response. Her cortical response was substantially attenuated when the stimulus (170-ms /da/ syllable) was presented in six-talker babble.
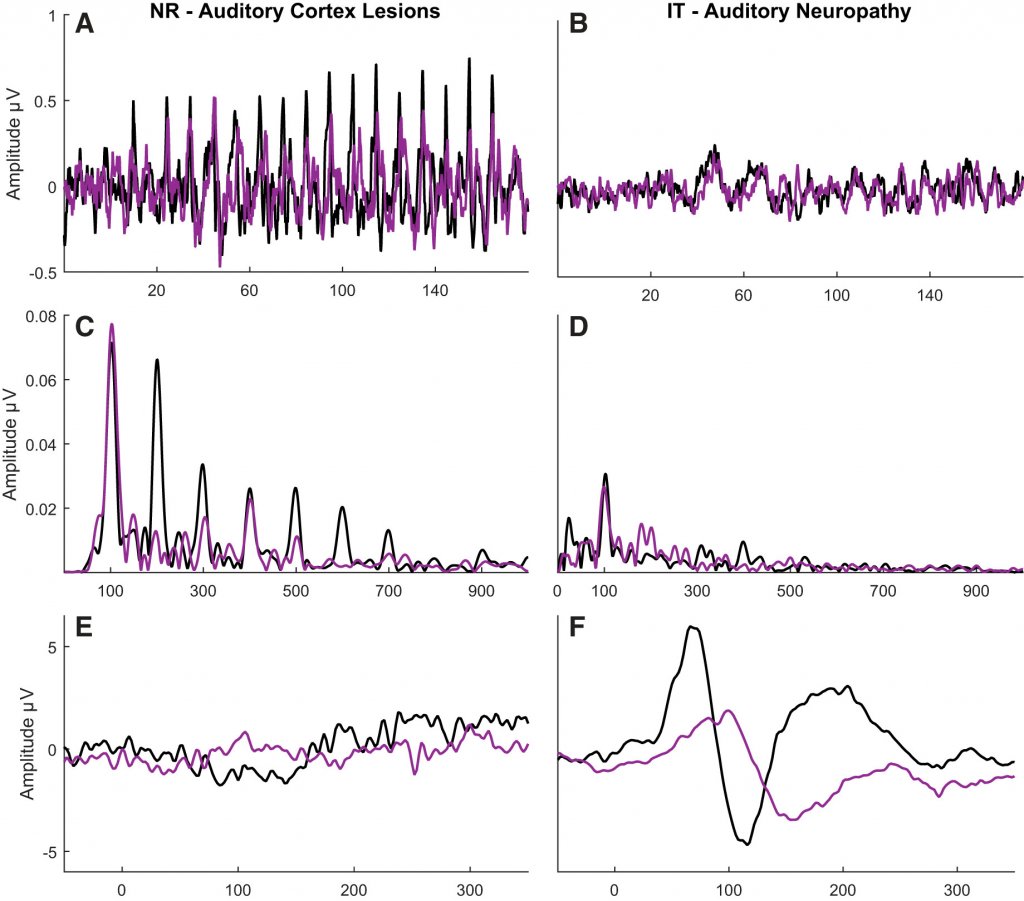
Figure 2. Contrasting frequency-following responses (FFRs) and cortical responses in an individual with cortical deafness (left panels) and auditory neuropathy spectrum disorder (right panels). The stimulus was a 170-ms /da/ syllable presented in quiet (black) or six-talker babble (purple). Panels A and B display the FFR response waveforms, and Panels C and D display the FFR frequency energy. Panels E and F display cortical response waveforms. Reprinted from White-Schwoch et al. Journal of Neurophysiology 2019.
These cases demonstrate the utility of using an array of evoked potentials to fully understand the nature of the patient’s complaints. Not every clinic/office has the equipment to provide these tests, but it would be important to remember that it is not possible to definitively conclude that a person has "normal hearing" based on an audiogram alone.
References
- Kraus N, et al. Consequences of neural asynchrony: A case of AN. J Assoc Res Otolaryngol 2000;01:33–45.
- Zeng FG, et al. Temporal and speech processing deficits in AN. Neuroreport 1999;10:3429–35.
- Anderson S, et al. Neural timing is linked to speech perception in noise. J Neurosci 2010;30(14):4922–26.
- Anderson S, et al. A neural basis of speech-in-noise perception in older adults. Ear Hearing 2011;32(6):750–57.
- Kraus N, et al. Auditory middle latency responses (MLRs) in patients with cortical lesions. Electroencephalograph Clin Neurophysiol 1982;54(3):275–87.
- White-Schwoch T, et al. Case studies in neuroscience: Subcortical origins of the frequency-following response. J Neurophysiol 2019.

